Clinical Examples of Gout
-
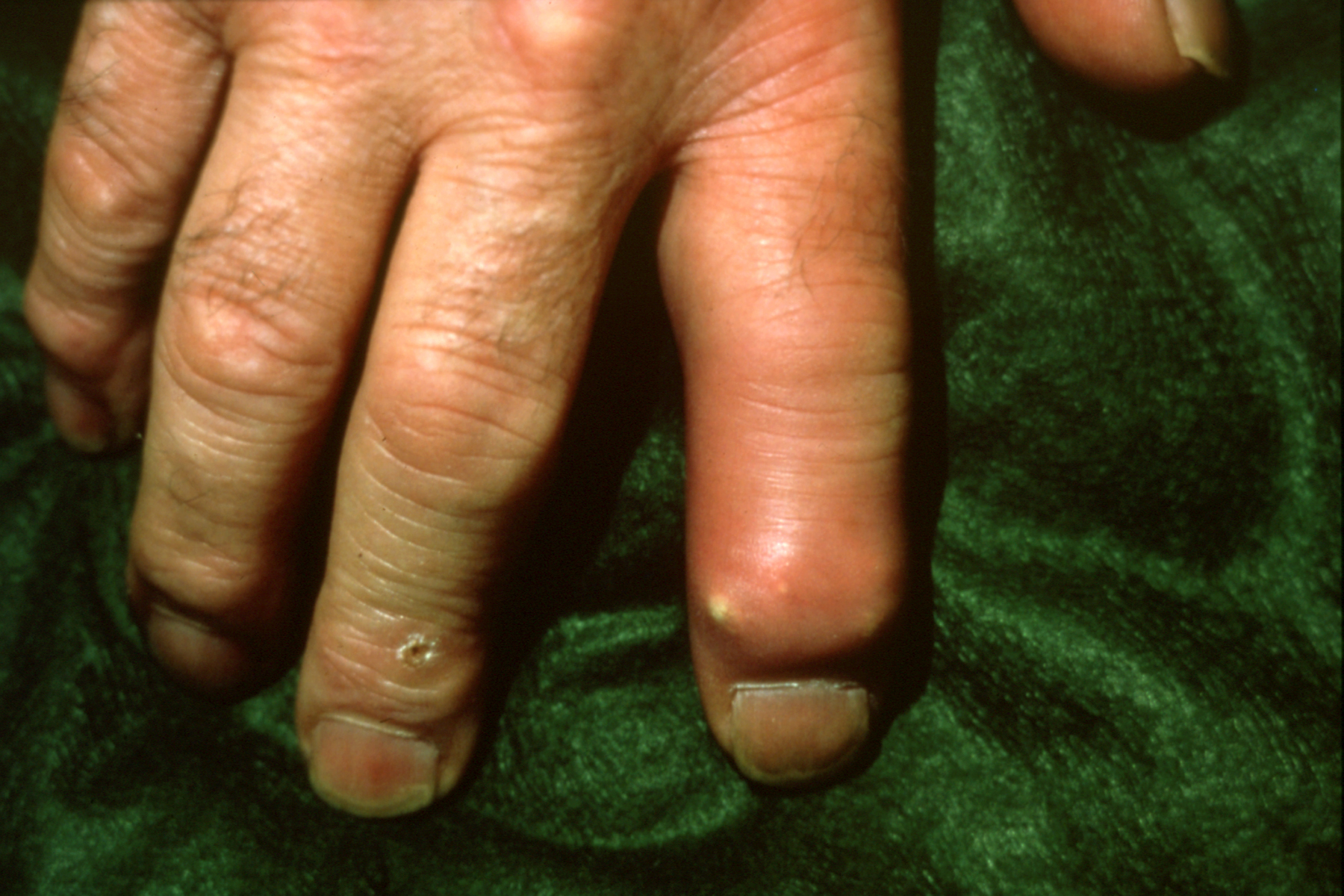
Gout at DIP presenting like a paronychia of the index finger.
-
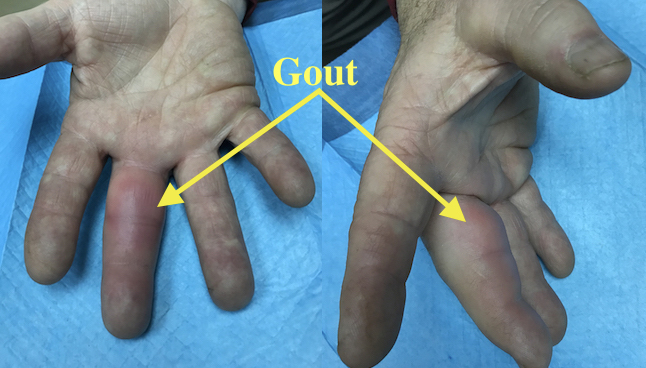
Acute Gout Long Finger causing pain and decreased range of motion
-
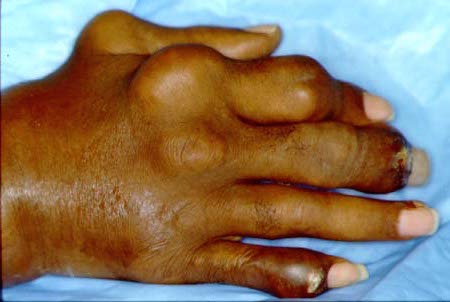
Severe uncontrolled chronic tophaceous gout
-
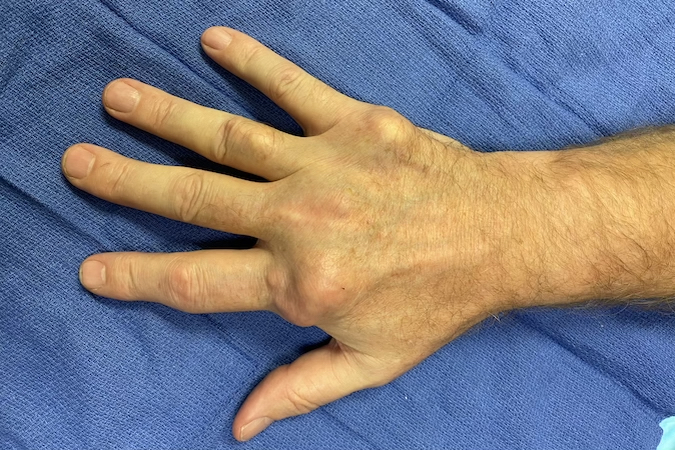
Tophaceous gout in right hand of a 57 year old male beer drinker.
